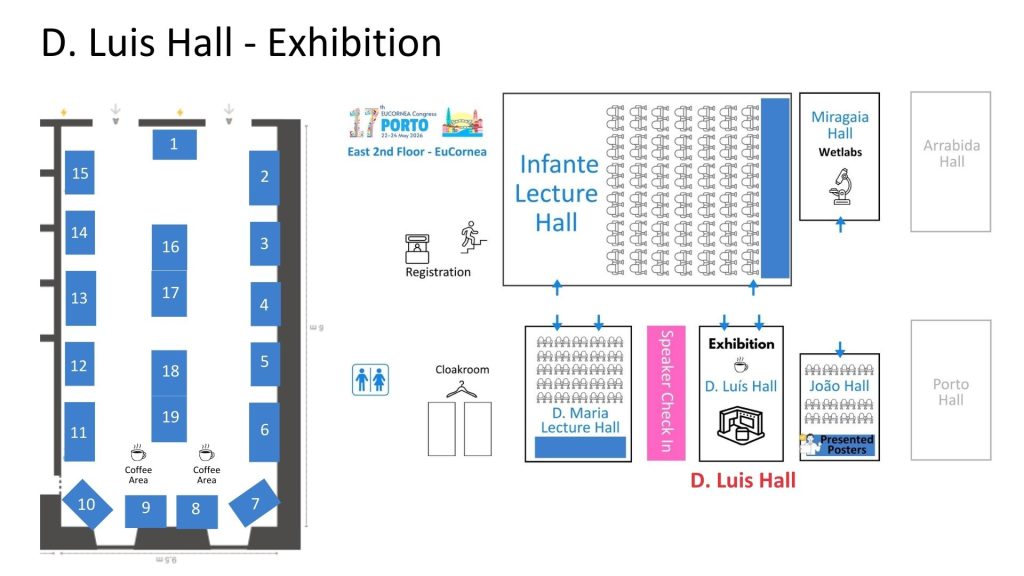
Exhibition Information
Menu
Be Aware of Unauthorised Agencies
You may be contacted by phone, fax, or email by third-party companies claiming to act as travel agencies or destination management companies. Some of these organisations use aggressive or deceptive tactics to solicit services or request information, particularly regarding hotels or congress registration.
Please note that the only official and valid links are those published on the Eucornea website: www.eucornea.org.

AJL Ophthalmic, S.A. is the only Spanish company (founded in July 1992) dedicated to the R&D, manufacture and marketing of medical devices in the field of Ophthalmology.
The company has reached a leading market position thanks to the fulfilment of very exigent Regulatory and Quality Control Requirements, 3000 m2 facilities with cutting-edge technology as well as the most qualified personnel for the manufacture of its products.
AJL has an extensive sales network with presence in more than 80 countries.
Our official website link. www.ajlsa.com

Our leadership is grounded in cutting-edge innovation and breakthrough technology, transforming the way we treat eye diseases and eye conditions. We have the widest array of eye care offerings in the industry with products organised into two businesses: Surgical and Vision Care.

Brill Pharma is a Barcelona-based pharmaceutical company focused on the research, development, production and commercialization of innovative healthcare solutions, with a strong emphasis on ophthalmology and surface ocular health. Founded in 2011, Brill Pharma collaborates closely with medical professionals and pharmacists to identify unmet clinical needs and translate them into high-quality therapeutic products designed to improve patient outcomes and quality of life.
Driven by a commitment to innovation, Brill Pharma places research and development (R&D) at the core of its activities, investing significant resources to generate scientific evidence and develop products with differentiated clinical value. This dedication to R&D ensures that new solutions are grounded in robust scientific principles and address real challenges in clinical practice.
With an experienced and multidisciplinary team, Brill Pharma has developed a range of products for ocular health and other therapeutic areas, always striving to support healthcare professionals with scientifically sound and patient-centred innovations. The company’s global reach and ongoing investment in R&D reflect its ambition to advance scientific knowledge and contribute meaningfully to the future of eye care.

Our exclusive CORONET® blade technology delivers exceptional precision and an ultra-sharp cutting edge, enabling clean, consistent trephination when it matters most.
Innovia Medical is a family of specialty surgical companies passionately committed to partnering with medical professionals to elevate patient care and improve clinical outcomes. Through focused innovation, engineering excellence, and close collaboration with clinicians, we help advance the standard of surgical performance.


Company Name: FIDIA
Company Email: info@fidiapharma.it
Company Website: https://www.fidiapharma.com/
Company Phone Number: Tel. (+39) 049 8232221 – 2222
Italian multinational company, with R&D, manufacturing and sales capabilities, and an extensive product portfolio mainly based on natural and functionalized hyaluronic acid (1,200 patents+), in joint care, skin care, eye care, specialty care and health&wellness care. Manufacturing operations, located in Italy, are inspected and approved by major international health authorities, including the US FDA, and the company extends its global reach through wholly owned subsidiaries and commercial partners in +120 countries worldwide.


Heidelberg Engineering pioneers imaging and data technologies to optimize ophthalmic solutions and support learning that advances research and empowers clinicians who want to improve the holistic health of patients. From its inception in 1990, the Company has collaborated with scientists, clinicians and industry to develop innovative products that deliver clinically relevant benefits.
Uncompromising quality and education play a large part in fostering the diagnostic confidence that has become synonymous with the global brand. The Company’s substantial expertise in the development and implementation of intelligent image and data management solutions complements its distinguished history in the design, manufacture and distribution of ophthalmic diagnostic instruments.
Heidelberg Engineering’s growing product portfolio combines these core technologies: confocal microscopy, scanning lasers and optics, optical coherence tomography (OCT), real-time image processing and analytics, multimodal image management solutions (PACS), electronic medical records (EMR) and large-scale data analysis.
The company’s ophthalmic diagnostic solutions and technologies are empowering clinicians to improve patient care in 120 countries, via 54 distributors.
Heidelberg Engineering GmbH
Max Jarecki Str. 8
69115 Heidelberg GERMANY
Phone: +49 6221 6463 0
Web: www.HeidelbergEngineering.com

Laboratoires KÔL was established in February 2020 to provide solutions to unmet needs in the field of ophthalmology, with a particular focus on corneal treatments and rare diseases.
A vocation to provide best in class pharmaceutical solutions to uncovered needs in the corneal field. An agile mindset to fast-track whenever possible product access for the benefit of patients. A team with a strong expertise in ophthalmic product development where every voice counts, listening to patients and doctors.
Our DNA :
KÔL is thus a new generation of pharmaceutical company : We are a French startup, at the heart of a dynamic entrepreneurial network in Clermont-Ferrand: one of the cradles of ophthalmology in France. We have an international ambition, and are proud and keen to export our French know-how.
Providing quality products that showcase French industrial expertise In-house expertise in the development of ophthalmic products. Partnering with selected public and/or private partners to boost our developments, create relevant synergies and increase our international footprint.
Innovative Products : Laboratoires KÔL currently markets two pharmaceutical products in eye drop solutions with Compassionate Access Authorisation. These derogatory authorisations are designed to make products available to patients to treat rare and serious diseases for which there is no therapeutic alternative.

MEDIPHACOS is a world-class company with over 50 years of experience in ophthalmology and presence in more than 60 countries in 5 continents. Our mission is to restore, preserve and improve human vision in collaboration with ophthalmologists through innovative, safe and effective products and services, like KERARING. KERARING are surgically implantable devices used to reduce corneal topographic irregularities, correct low and high-order optical aberrations and improve visual acuity in patients with keratoconus and other ectatic disorders. KERARING was specifically designed to treat irregular corneas and has been the treatment choice for hundreds of thousands of patients globally for over 20 years with excellent safety, efficacy and low complication rates.
Email: info.europe@mediphacos.com
URL: www.mediphacos.com

Nordic Pharma is an international pharmaceutical and medical device company with a history of internal product development and acquisitions.
Through a combination of innovation, collaboration and quality, Nordic Pharma strives to address specific unmet medical needs.
Nordic Pharma holds an established position in Women’s Health, a fast-growing Rheumatology franchise with a global footprint, and an increasing international presence in Ophthalmology.
We bring over 30 years of expertise since our creation in 1995.

Since 1895 OCULUS has been a trusted partner for eye care professionals around the world. With the highest attention to detail, our devices are designed and manufactured at the OCULUS headquarters in Germany.
Stop by our booth to find out why it is clever to integrate the new Pentacam® Cornea OCT to your practice. Test the latest OCULUS device and talk to our product specialists – we are here for you.

Optico Ltd is a British manufacturer with decades of expertise in precision ophthalmic microsurgical instrumentation. Trusted by surgeons worldwide, they combine traditional craftsmanship with modern engineering to deliver instruments that offer exceptional control, balance and reliability in the operating theatre.
Working in close partnership with leading ophthalmologists, Optico continuously refines and develops solutions that support the evolving needs of corneal, cataract and vitreoretinal surgery. As their international presence expands, the commitment remains clear: precision without compromise, innovation driven by surgical insight and instruments designed to help surgeons achieve consistently outstanding patient outcomes.
For more information visit www.optico.org.uk

SIFI, leading ophthalmic company, headquartered in Italy, since 1935 develops, manufactures and markets innovative therapeutic solutions for patients with ophthalmic disorders. SIFI portfolio includes innovative pharmaceuticals, medical devices and food supplements exported to more than 60 countries worldwide.
SIFI has direct commercial operations in Italy, France, Spain, Romania, Turkey and Mexico.
In 2022 SIFI has entered into an agreement to establish a Joint Venture to develop, manufacture and commercialize premium innovative intraocular lenses (“IOLs”) in the Greater China market, including Taiwan, Macau and Hong Kong and the United Arab Emirates in 2024.
In October 2024, Akantior, the new drug for Acanthamoeba Keratitis ultra-rare corneal disease, has been launched in Germany. It’s going to be launched in Italy and in US.
Akantior is also going through the regulatory approval procedures for fungal keratitis.

Tarsus Pharmaceuticals, Inc. applies proven science and new technology to revolutionize treatment for patients, starting with eye care. Tarsus is advancing the development and commercialization of therapeutic candidates to address highly prevalent diseases with limited treatment options across a range of therapeutic categories including eye care and infectious disease prevention. Their lead product, XDEMVY® (lotilaner ophthalmic solution) 0.25%, formerly known as TP-03, is FDA-approved in the United States for the treatment of Demodex blepharitis but has not yet been approved in Europe.
Tarsus is on-track for potential European regulatory approval in 2027 of a preservative-free formulation of TP-03. Tarsus is also developing TP-04 for the potential treatment of Ocular Rosacea and TP-05 for the potential prevention of Lyme disease.
Learn more at TarsusRx.com.

Théa is a profoundly European company whose independence, stability and entrepreneurial spirit are safeguarded by the Chibret family, which has been instrumental in the field of ophthalmology for over 150 years. Being a family-owned business has allowed Théa to develop over the long-term, focusing on employees and human values to fulfil our mission of being ‘The’ ophthalmology specialist. A pioneer in preservative-free treatments, we are committed to providing healthcare professionals and patients with innovative products and a wide range of specialty items (service products, daily-use products) across all ophthalmological therapeutic classes.
We are continually reinvesting in our two main areas of interest, Innovation and Internationalisation, with long-term strategy being favoured over immediate profit. Our goal is to pursue and strengthen lasting partnerships and collaborative research, to share our expertise and address unmet medical needs to provide new methods for treating and supporting patients. Internationalisation is in our genes: currently the leading independent eye care group in Europe, Théa has set its sights on becoming a world leader by continuing to export its products and establish itself internationally, while always respecting the cultures of each country.
Sponsorship Opportunities
A tabletop exhibition will be an integral part of the Porto Congress. The exhibition will be conveniently located next to the lecture halls, allowing company representatives to engage directly with delegates.
There are also many sponsorship opportunities available to help you promote your company’s visibility and brand.
To receive a copy of the Exhibition & Sponsorship Prospectus Brochure & Booking Form, please contact: eucornea@eucornea.org